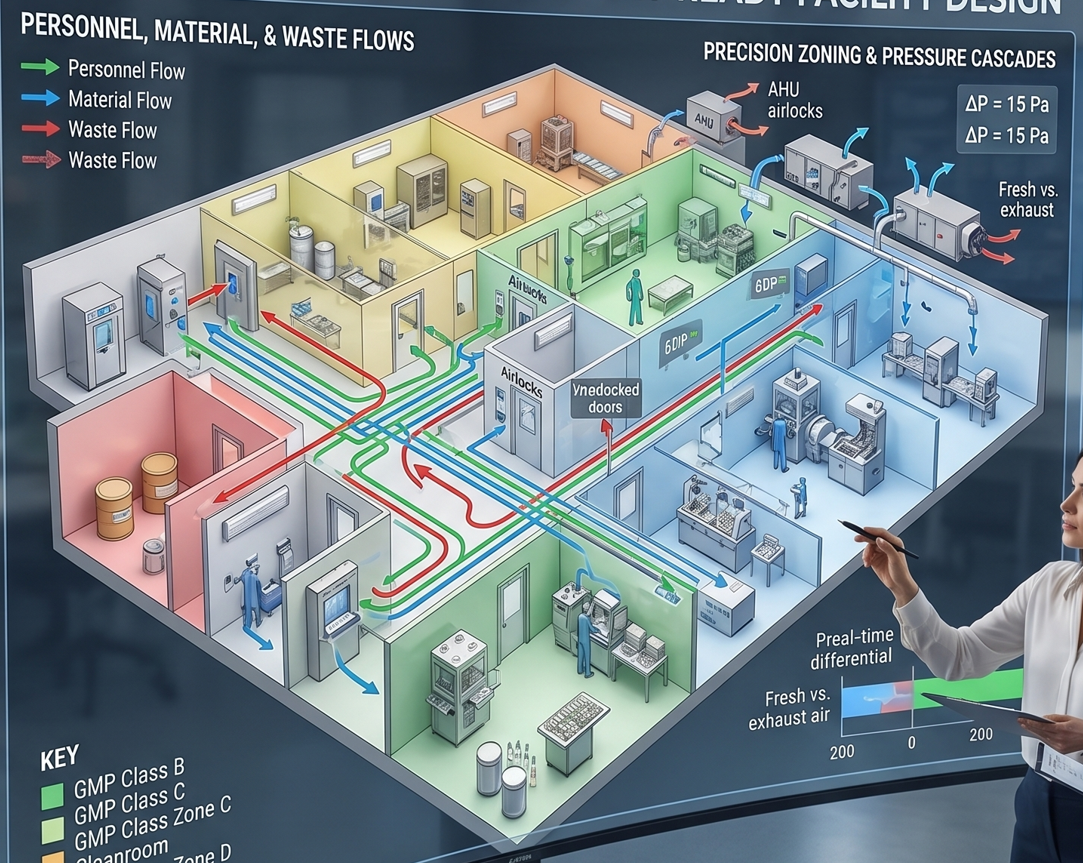
Precision Engineering for Critical Environments.
Specialized HVAC advisory and conceptual design for GMP-compliant pharmaceutical cleanrooms and high-capacity industrial extraction.
The Difference Between "Pretty" and "Compliant"
Many design firms can render a colorful, eye-catching 3D floor plan that looks impressive to an investor. But an experienced pharmaceutical engineer will spot the GMP audit failures immediately.
Look closely at the conceptual layout below. Can you spot the critical GMP violations?
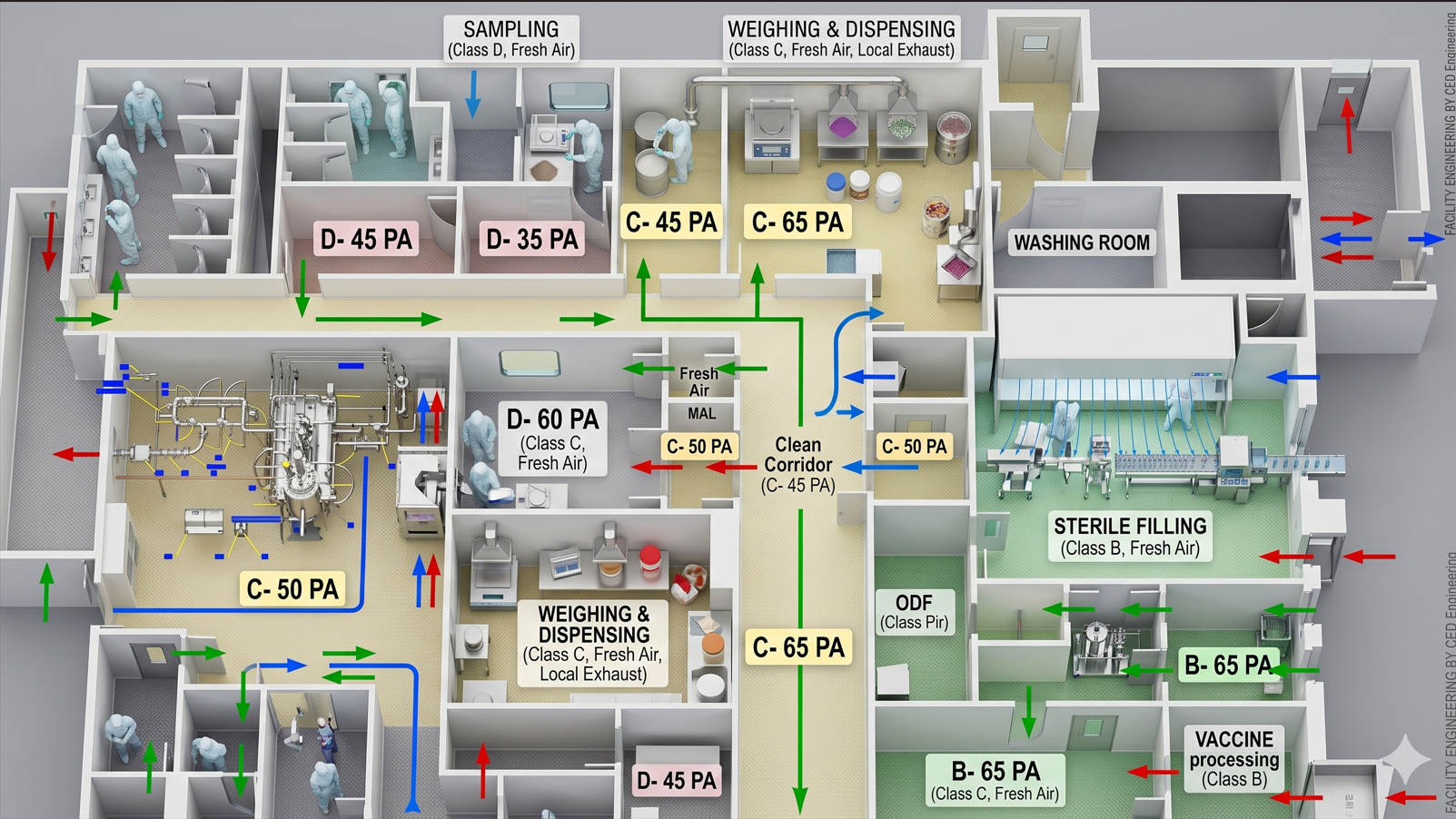
The Engineering Reality Check
Fatal Pressure Cascades: This layout shows a Class D zone running at 60 PA next to a Class C zone at 50 PA. In a positive-pressure facility, this forces contamination backward into the cleaner zone.
The Missing Airlock (PAL/MAL): Flow arrows indicate personnel moving directly from a Class C corridor into a Class B sterile filling core. There is no dedicated gowning step-up or air shower transition bridging the gap—an immediate compliance failure.
SOP & Gowning Violations: Personnel wearing full Class B sterile hoods are depicted operating inside lower-grade Class D washing and sampling areas, completely destroying the sterile integrity of the garments.
Washing Room: should have been divided into Dirty, Washing, Drying and Clean.
Corridor Classification & Transitions: The main clean corridor should be designated as Class D. Accessing the Class C rooms requires secondary change rooms (CH.Rs) and proper PAL/MAL step-ups, rendering the depicted personnel flow strictly non-compliant.
At CED-Engineering, we don't just draw pictures. We engineer reality.
We provide airtight, audit-ready facility layouts to ensure your facility passes inspection the first time.
Expert Consulting Solutions for Business Growth.
Expert Consulting Solutions for Business Growth.
Our Services
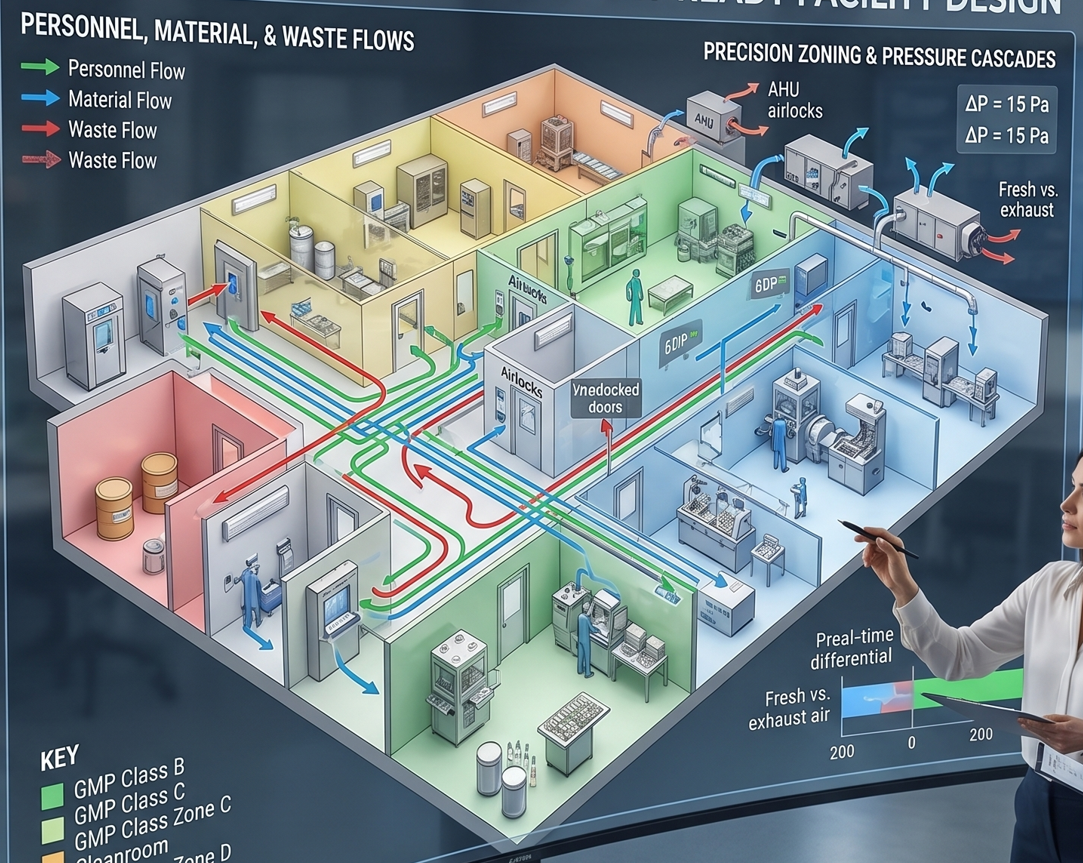
Over Two Decades of GMP-Compliant Facility Engineering Leveraging 20+ years of specialized experience in pharmaceutical manufacturing, CED Engineering provides comprehensive, audit-ready facility layouts. We design critical environments tailored to your specific manufacturing lines, including powders, solid dose (soft/hard capsules), sterile injectables (ampoules and vials), ODF, and vaccines across Class B, C, and D cleanroom standards.
Comprehensive Layout & Flow Strategy:
Strict Contamination Control: Advanced mapping of personnel, material, and waste flows to eliminate cross-contamination risks.
Precision Zoning & Pressure Cascades: Strategic HVAC integration detailing pressure differentials, cleanroom zoning, and exact fresh air versus exhaust air matrices.
Complete Spatial Architecture: Seamless, compliant integration of all critical nodes, including specialized airlocks, weighing/dispensing rooms, preparation areas, filling lines, and packaging zones.
Engineering for Constructability, Maintenance, and Energy Efficiency We do not just design systems on paper; we engineer them for operational reality. Backed by extensive field experience in industrial commissioning, QA/QC, and facility management, CED Engineering delivers robust, energy-efficient HVAC architectures. We bridge the gap between theoretical design and heavy-duty operation, ensuring your cleanroom systems are not only strictly GMP-compliant but highly maintainable and built for a maximum lifecycle.
Core HVAC & Mechanical Capabilities:
Constructability & Lifecycle Engineering: Systems designed with the facility manager in mind. We prioritize maintenance access, seamless QA/QC validation, and durable component selection to drastically reduce long-term operational downtime.
High-Efficiency Thermal & Control Systems: Advanced integration of variable speed drives (VSD), precision inverters, and optimized air handling units (AHUs) to minimize energy consumption without ever compromising critical environmental parameters.
Strict Environmental Control: Precise load calculations for air change rates (ACR), terminal HEPA filtration routing, temperature/humidity tolerances, and differential pressure mapping to guarantee airtight Class B, C, and D compliance.